The 7 Sins of GMO Testing

The use of genetically modified (GM) crops is increasing around the world. This presents many opportunities to the agricultural industry while it brings many challenges to the analysis industry. One Romer Labs expert shares his knowledge about testing GM crops so that you can avoid making common mistakes in your laboratory.
1. Identifying the wrong target
Do your homework: this is perhaps the most basic habit any scientist can learn. This is particularly necessary for genetically modified organism (GMO) testing, where doing your homework means knowing which events comprise your targets so that you can avoid false positives and erroneous results.
The problem relates to traits. A trait is a protein derived from a genetic modification which gives a special feature to the plant. Genetic modifications may be present in different combinations that produce similar—or entirely different—traits. Among the most popular elements of genetic modifications are promoters (p35S, FMV), terminators (NOSt), coding genes for certain useful traits (cp4 epsps, pat, bar, Cry1A, and others), and genes coding selective markers (NPTII, PMI).
Your technicians need to consider many different factors in defining the target for any GMO analysis. Make sure you consider the territory the product originates from as well as the possible events, whether these events are authorized in the region in question, and whether the plant of interest has commercially available biotech traits.
It can be tricky: sometimes the modification exists but is not authorized or planted in certain regions. However, it may be widely used in another region. In addition, there have been cases of unapproved events escaping containment and making their way to the field.
2. Choosing a faulty or insufficient method
It can be a risky decision: what should be the scope of any GMO screening? Laboratories can narrowly target a few genetic elements or proteins, or they can expand the scope to several events or proteins, which can be expensive. Finding the method that fits your needs and ensures accurate results is essential.
DNA-based methods are time-consuming and depend on high-quality DNA extraction and proper controls. Copy number variation and polyploidy can also cause problems in samples and should be taken into consideration when performing DNA analysis.
A look at some common plants will illustrate the point. In the case of soybeans, it is not enough to test only for promoters and terminators, because only a few of the 15 potential soybean events contain these common genetic elements. Maize is a different story altogether: broadly screening for these elements is almost completely sufficient, because most events in maize contain these elements.
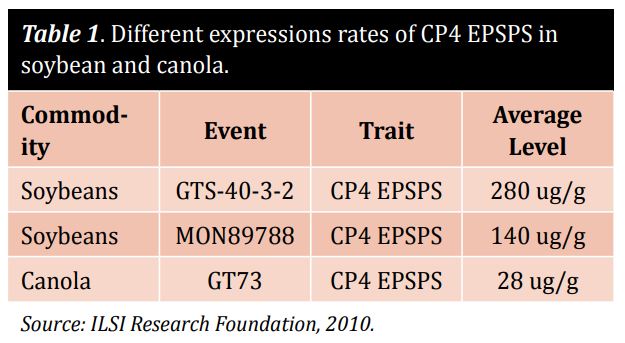
While DNA-based methods are expensive and used only in analytical service labs, protein identification is relatively cheap and is useful in screening for common traits on-site. The majority of GM events has its own level of modified protein expression. Be careful when carrying out protein analysis of soybean and canola with the same lateral flow device (LFD) strips. Table 1 highlights different expression rates of CP4 EPSPS, making it necessary to use different LFD detection methods in soybean and canola. The right approach uses different tests and adjusts the sensitivity for different commodities to reach a similar level of detection.
3. Not fully understanding different degrees of test sensitivity
DNA-based methods are very sensitive and are able to detect a single copy of the target gene. The sensitivity for DNA-based methods is limited by the number of seeds tested. It is usually 0.01% or one genetically modified (GM) seed in a set of 10,000 non-GM seeds. Such sensitivity is more than enough to be 99.9% sure of any specific level of detection down to 0.1%.
On the other hand, protein-based methods have a limit of detection (LOD) as low as 0.1%, or 1 GM seed in 1,000 non-GM seeds. Yet this sensitivity is not enough to guarantee a result with 99% certainty. The best you can get with a single protein test with an LOD of 0.1% is a level of 0.46% at 99% confidence. The non-uniform distribution of the seeds in real samples leads to this discrepancy. To be 99% certain that the level of GMO contamination is lower than 0.1%, we recommend that you carry out at least four tests of 1,000 kernels at a sensitivity of 0.1% each. For 95% confidence at 0.1%, carry out three tests of 1000 kernels all with negative results.
4. Removing impurities or treating the sample
Grain labs often remove impurities from samples before conducting a test. However, the purification of GMO samples is not allowed. If some residues containing traces of GMO plants are left in the samples, they should be detected. For example, if a corn sample contains some residues of GMO soy (kernels, shells, dust), washing and manual cleaning will remove these residues and produce an incorrect result.
Do not subject the samples to any kind of heat treatment, not even in order to dry excess moisture. For protein-based methods in particular, this could result in the denaturation of the trait proteins and lead to the complete loss of test sensitivity, as the target protein would have changed. Antibodies used in these applications would then no longer be able to recognize their targets.
5. Using an improper procedure to prepare the sample
Your method of DNA isolation should be carefully designed and followed to avoid DNA degradation, which could skew your results. Therefore, it is important to verify the quality and quantity of purified DNA. In addition to the DNA extraction reagents used, the grinding method is also very important. Blade mills give the best results, as they grind finly enough to enable DNA extraction. However, it is imperative to thoroughly clean any grinding equipment after use, as cross-contamination is a huge problem in GMO testing. Equipment that is not properly cleaned could produce false negative results in subsequent batches.
Protein extraction requires a coarser grind. It is enough simply to crush all seeds in the sample. If you grind seeds too finely, it will take too long for the extract to settle, and the sample will contain too many solid particles, which will stick to the LFDs and inhibit the flow of liquid. Mills used for extraction before LFD analysis should be set to avoid causing fractions that are too fine. The shape of the extraction vessel should also be considered: it should not be flat, and it is better to use disposable vessels to avoid cross-contamination. Blenders are very commonly used for grinding for protein testing because they are very easy to clean, therefore avoiding problems with cross-contamination.
6. Customizing testing procedures or failing to read the package insert
This could well be the most serious sin that operators commit. While reference testing labs employ trained staff throughout the whole year, grain labs often have a seasonal employment policy and therefore employ staff with less expertise in testing.
This could lead to the testing procedure being misread, incorrectly followed or altered in the lab. This can affect all parts of the testing process, beginning with extraction and affecting testing elements such as the volumes of liquid required for the test, development times, and analysis procedures.
Make sure you thoroughly read all packaging and documentation before carrying out any analysis to ensure reliable results.
7. Insufficiently cleaning your equipment (causing cross-contamination)
Make sure that the mill you use for GMO analysis is easy to clean. The best way is to use removable jars and blades so that you can clean them separately from the mill motor. Every component of your equipment should be washable. Blenders are commonly used for grinding GMO samples as they are easy to clean. Mills have to be dismantled and all the parts washed separately.
Wash all equipment with liquid soap and rinse with water afterwards. Do not use ethanol as a cleaning agent; its only use is to accelerate the drying process. Each lab should validate its cleaning procedure to ensure efficiency.
We recommend the use of gloves when handling all samples. Changing your gloves and washing your hands between samples will also help to avoid cross-contamination.
Make sure you clean any tools that may have come into contact with the extract of ground material. Another solution is to use disposable equipment as much as possible.
Published on:
GMO