GMO Testing: 4 Important Questions

Are you testing protein or DNA? How do methods for testing raw materials differ from those of processed materials? These are just two considerations that anyone looking to test for GMOs needs to keep in mind. Product manager Christina Huber offers some practical answers.
As discussed in the previous article, different countries have different regulations regarding approvals for trading with genetically modified plants or cultivating them within their jurisdiction. Hence, the criteria for testing vary significantly among countries. This article lays out some basic considerations when choosing the right test format to determine whether a sample is derived from a genetically modified plant. Genetic modification refers to any artificial alteration of the DNA in a plant, either by editing the genome or by inserting foreign fragments of DNA. The modified gene will convey a new trait, such as insect resistance, herbicide resistance or other agriculturally relevant features. One of the first questions that you will encounter is whether you want to test DNA or protein. Complementary to this decision, you have to decide whether the raw commodity or processed material will be tested. A third matter is your target: are you looking for a specific GMO trait, or do you simply want to know whether your crop is GMO-free? Finally, factors such as sensitivity, speed and ease-of-use play a large role as well. All these decisions will help you in choosing the right format from the most commonly applied testing options: polymerase chain reaction (PCR), enzyme-linked immunosorbent assay (ELISA) or lateral flow devices (LFD).
1) DNA or protein?
As the plant is being modified, its DNA sequence is changed. The alteration can be determined by identifying the artificial alteration of the DNA itself or by detecting the expressed protein, which does not occur naturally in the crop in question.
Nucleic acid-based testing approaches like PCR amplify a region of interest of the plant genome to obtain billions of copies of small DNA fragments using so called primers and DNA polymerase as enzyme. Previously, those DNA fragments could be separated and visualized on an agarose gel but nowadays more sophisticated instruments allow for the direct measurement of the amplification reaction based on fluorescence techniques. Not only is a PCR approach qualitative, it also allows for the quantification of GM material in a sample.
For the detection of proteins, immunoassays are used. These immunological tests use specific antibodies that can bind the protein. Two different testing formats are available – lateral flow device (LFD) and enzyme-linked immunosorbent assay (ELISA). Both systems work in a sandwich-format, meaning that one antibody is immobilized on a surface that specifically binds the protein of interest. Once the protein has bound to the immobilized antibody, a labeled secondary antibody can bind, thereby forming a sandwich and giving a color signal. If this protein of interest is not available, no sandwich can be formed and therefore no color will be visible, indicating that the tested plant is GMO-free. In most cases, ELISA uses a 96-well plate coated with the specific antibody that allows for the simultaneous analysis of multiple samples. LFDs are strips that have a test and a control line coated with the specific antibody to capture the protein of interest. If the test line appears, the result is positive, indicating the presence of the target protein. These tests exist in a quantitative or qualitative format; an LFD reader is required to quantify the result. In sum, DNA testing will read the altered DNA sequence, whereas protein testing identifies the new protein derived from this specific DNA sequence. The specific qualities of the sample to be analyzed also plays a role in determining whether a DNA-based or protein-based method best fits your needs. Moving on, we will look at the different requirements for testing raw commodities or processed materials.
2) How does testing for raw materials differ from that of processed materials?
When it comes to determining the appropriate testing technique, a further question beyond the target (DNA or protein) arises: does the matrix consist of raw or processed material? This is particularly relevant for protein testing. Raw materials express the intact protein, meaning that both PCR and immunological tests can be used. However, once a raw material has been processed, its proteins denature with the result that they change their natural structure and, in most cases, cannot be detected by immunological tests. Heat treatment, for example, is a process that can lead to the denaturation of proteins. However, this process does not change the DNA sequence; therefore, PCR can be used to detect the inserted gene even in some processed materials.
More testing options are open to you if your sample is a raw commodity: both DNA- and protein-based approaches will detect the genetic modification. If, however, your sample is a heavily processed material in which the protein may have been denatured, your options are restricted to DNA testing.
3) Is there a specific trait in my sample? Is it GMO-free? What exactly do you need to know?
Sometimes, it is necessary to test a sample for specific traits in order to determine whether it contains plants not permitted in certain regions. It may also be necessary to determine whether the sample is completely free of GMOs, regardless of any particular trait. To test for the presence of a specific GMO, it is important to know which proteins are expressed in the GMO of interest. This information can be acquired from the producer or from sources such as the ISAAA webpage (http://www.isaaa.org/), where all approved GMOs are listed.
The next step is to determine the desired test format (PCR, ELISA or LFD) based on the desired specificity, time-to-result, type of material, and laboratory equipment available. Analysts need to be careful here, as there are many GMO products that contain not just one inserted gene but several (called stacked events); therefore, if you want to test for a specific GMO, you need to test for all inserted genes (or expressed proteins) associated with that particular plant.
Another strategy for GMO testing is to determine whether the plant contains any GMO materials at all. This type of testing is not specific to any particular event. To detect whether a sample is completely free of GMOs, it is sufficient to test only for one event of each crop; this is effective even considering GMO crops with stacked events, as proving the presence of only one event is sufficient to prove the presence of a GMO. All three techniques are appropriate for this kind of test. PCR would provide the widest range, as, for example, the NOS-terminator or 35S promoter is very often inserted. When testing with LFD strips, a combinational strip can detect several proteins at once. These two approaches depend solely on the needs of the customer, as for some it might be more important whether the crop is GMO-free (as in a country were cultivation is banned but illegal planting might occur). Others might want to know specifically which plant they have been planting.
4) How can you choose the right testing method when taking other factors such as the testing environment into account?
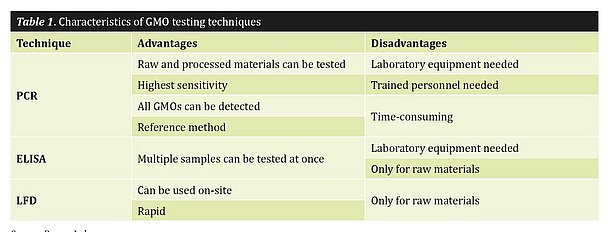
Another factor that plays a role in choosing the right testing format is the testing environment. Will you have a laboratory and trained personnel available or would you rather have a quick test that does not require any laboratory equipment? Table 1 summarizes the available testing formats and their advantages and disadvantages.
Let’s go through the table step-by-step. As the reference method for GMO testing, PCR is the most suitable method if you want to test any processed material. This technique also provides the highest degree of sensitivity and detects virtually any GMO. However, one significant drawback is that laboratory equipment and trained personnel are needed. To perform a PCR, especially when quantification is desired, exact pipetting skills are essential, because imprecise pipetting could lead to wrong or inconclusive results.
The major advantage of an ELISA is the ability to test multiple samples simultaneously. This is important when you want to carry out single-seed testing. However, as this is a protein-based test, it can only be used for raw materials.
If you compare all three techniques in terms of time-to-result, LFD is the fastest method. LFD strips can be used on-site and do not require sophisticated instrumentation. So, if you want to test an incoming shipment and need fast results, it would be very reasonable to choose an LFD test, as results are available within 10 minutes; no laboratory personnel and little equipment are needed. This technique also does not require any laboratory skills, as the procedure is designed to be easy to use.
So which method works best? Your decision is ultimately a function of your desired time-to-result and the facilities, equipment and personnel at your disposal.
Published on:
GMO
Conclusion: the value of customized solutions for GMO testing
All in all, you must take into account several factors to find the right testing strategy. It depends on the material that you want to test (raw or processed), whether you need fast results, whether you have the capacity to set up a laboratory with equipment and personnel, and whether you want to know specifically which GMO trait you have or if you want to know whether your crop is GMO-free. There is no right or best technique, as your decision depends on your answer to all these questions. Some may choose LFDs in order to meet tight deadlines, whereas others may want more specific and detailed results using PCR. Still others, such as research institutions, may want to test their research-trial field and do single-seed testing of multiple samples using ELISA. Taking all these elements into consideration will help you make the right choice when it’s time to test your crop.